Hi guys,
One of the primary concerns for these LED lights is the thermal relief of the heat generated by the LED. We know this and have discussed it to a great extent! In a recent run time test on a driver/ battery conbination which I felt was pushing the power limit and after touching the hot light during the test, I decided to try a run time test with the light sitting in a pan filled with a significant volume of water. I reasoned that the water would provide more than ample thermal relief to the LED. This was the case as the head was not even warm to the touch during a subsequent run time test. However, the results of the test were disconcerting as the timein regulation was significantly shorter than the previous test! This has been in the back of my mind and bugging me ever since. During the last week or so, I have run a number of air and then water cooled runtime tests on various lights.
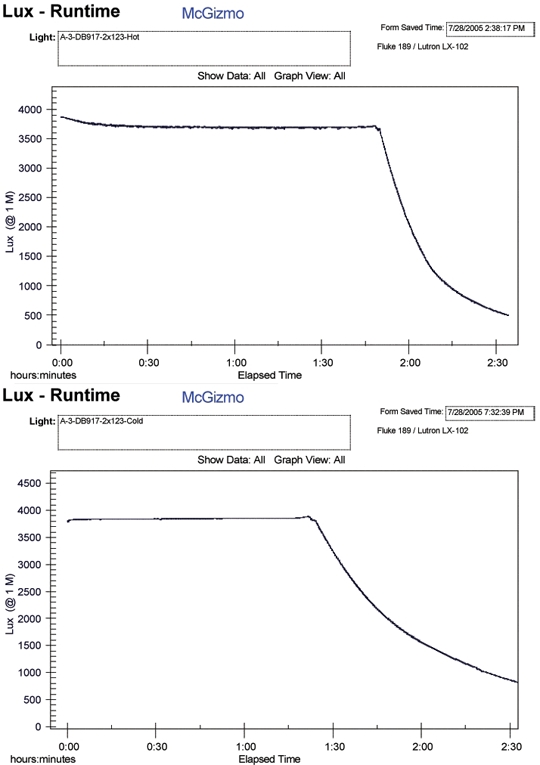
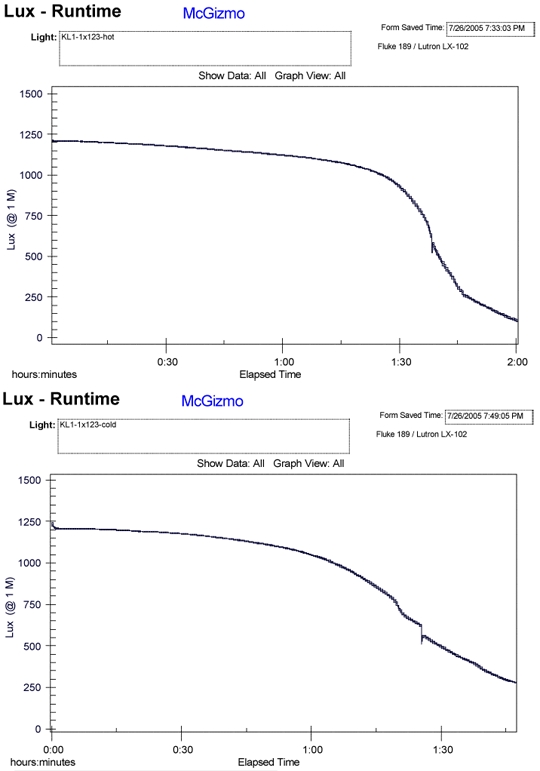
In each pair of tests, the air as well as water has been ambient and the same, within a few degrees. I have combined a few of these air/water (Hot/Cold) tests and put them on a page, HERE . Below I will include a couple of the runtime paired graphs as well. In all cases, there is a significant and noticible reduction in the primary lithium cell's capacity when it is in the water cooled environment!!!
I was under the impression that one of the real advantages to lithium chemistry is the consideration that these cells work well in cold applications. I am now of the opinion that they may work but at potentially diminished capacities!! perhaps this information has been known and discussed in the battery and electronic forums but I sure haven't picked up on the fact!! /ubbthreads/images/graemlins/icon15.gif It would seem that what is ideal for the LED (25C) is not ideal for its energy source?!?!? /ubbthreads/images/graemlins/icon3.gif From the LED manufacturers, we have cool graphs of flux VS temperature. Are there similar graphs matching a cells capacity VS temperature? With a low thermal resistance in a flashlight, I would assume that at some low ambient air temperature that the lithium battery would perform in a diminished capacity?!? Do our lithium based flashlights need body warmers in the winter time?!? /ubbthreads/images/graemlins/thinking.gif
**********
**********
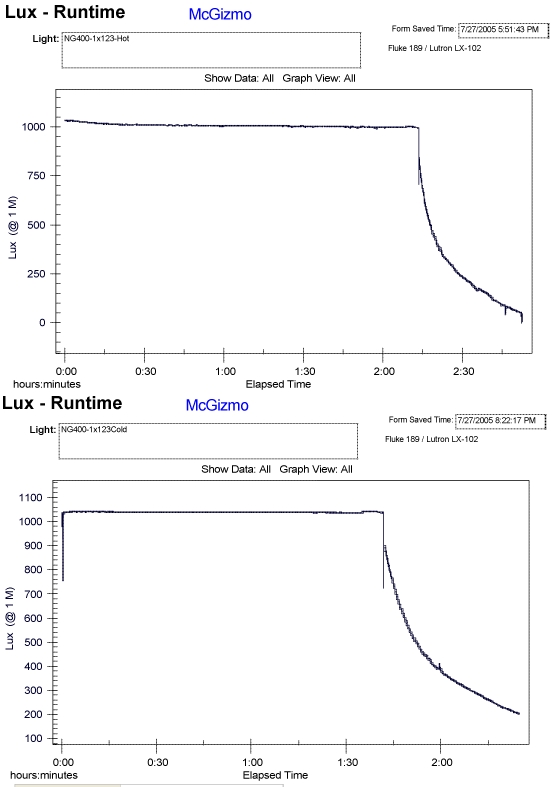
There are a few more examples shown on the linked page above but I think the trend is rather clear. It would seem that not only is the capacity (amp hours) diminished but the ampacity or max power available is also diminished. I did one test shown with a Ti head and body and I figured that the thermal resistance of the metal would give the battery a better chance of remaining at an elevated temperature. It may well have helped but the reduction in run time was still way too significant and over shadows any hint of aid.
All of the CR123 cells used in the test were SF and the CR2's usd in the test were from Battery Station. I have no idea if this evidence is based on the general chemistry of lithium primaries and for all I know the fact that the batteries were standing on end may be contributory. I do know that I am ignorant and possibly stupid in regards to these tests and their results. I trust some of the battery gurus can enlighten us!?!?!
I mentioned this to Wayne Y. the other day and he suggested that this thermal "dependency" might also have some bearing on the noted results of two batteries in series having different discharge rates. /ubbthreads/images/graemlins/thinking.gif /ubbthreads/images/graemlins/icon3.gif
So, in short, what's the skinny here and do I need a neoprene/ down bootie for my light if I take it underwater or up in snow country?!?! /ubbthreads/images/graemlins/grin.gif
I am also curious as to how the delta in C will vary for the Li-Ion cells as a function of T. This last statement almost makes it sound like I know what I am talking about! /ubbthreads/images/graemlins/crackup.gif
One of the primary concerns for these LED lights is the thermal relief of the heat generated by the LED. We know this and have discussed it to a great extent! In a recent run time test on a driver/ battery conbination which I felt was pushing the power limit and after touching the hot light during the test, I decided to try a run time test with the light sitting in a pan filled with a significant volume of water. I reasoned that the water would provide more than ample thermal relief to the LED. This was the case as the head was not even warm to the touch during a subsequent run time test. However, the results of the test were disconcerting as the timein regulation was significantly shorter than the previous test! This has been in the back of my mind and bugging me ever since. During the last week or so, I have run a number of air and then water cooled runtime tests on various lights.
In each pair of tests, the air as well as water has been ambient and the same, within a few degrees. I have combined a few of these air/water (Hot/Cold) tests and put them on a page, HERE . Below I will include a couple of the runtime paired graphs as well. In all cases, there is a significant and noticible reduction in the primary lithium cell's capacity when it is in the water cooled environment!!!
I was under the impression that one of the real advantages to lithium chemistry is the consideration that these cells work well in cold applications. I am now of the opinion that they may work but at potentially diminished capacities!! perhaps this information has been known and discussed in the battery and electronic forums but I sure haven't picked up on the fact!! /ubbthreads/images/graemlins/icon15.gif It would seem that what is ideal for the LED (25C) is not ideal for its energy source?!?!? /ubbthreads/images/graemlins/icon3.gif From the LED manufacturers, we have cool graphs of flux VS temperature. Are there similar graphs matching a cells capacity VS temperature? With a low thermal resistance in a flashlight, I would assume that at some low ambient air temperature that the lithium battery would perform in a diminished capacity?!? Do our lithium based flashlights need body warmers in the winter time?!? /ubbthreads/images/graemlins/thinking.gif

**********

**********

There are a few more examples shown on the linked page above but I think the trend is rather clear. It would seem that not only is the capacity (amp hours) diminished but the ampacity or max power available is also diminished. I did one test shown with a Ti head and body and I figured that the thermal resistance of the metal would give the battery a better chance of remaining at an elevated temperature. It may well have helped but the reduction in run time was still way too significant and over shadows any hint of aid.
All of the CR123 cells used in the test were SF and the CR2's usd in the test were from Battery Station. I have no idea if this evidence is based on the general chemistry of lithium primaries and for all I know the fact that the batteries were standing on end may be contributory. I do know that I am ignorant and possibly stupid in regards to these tests and their results. I trust some of the battery gurus can enlighten us!?!?!
I mentioned this to Wayne Y. the other day and he suggested that this thermal "dependency" might also have some bearing on the noted results of two batteries in series having different discharge rates. /ubbthreads/images/graemlins/thinking.gif /ubbthreads/images/graemlins/icon3.gif
So, in short, what's the skinny here and do I need a neoprene/ down bootie for my light if I take it underwater or up in snow country?!?! /ubbthreads/images/graemlins/grin.gif
I am also curious as to how the delta in C will vary for the Li-Ion cells as a function of T. This last statement almost makes it sound like I know what I am talking about! /ubbthreads/images/graemlins/crackup.gif

