Well op, now you know why you had to do so much reading. Because there are some people who work very hard to spread false information that they think is true. It is an ego thing,
Blaming and the passive aggressive behavior seen in his previous posts are common symptoms of JustAnOldFashionedLEDGuy's mental illness, NPD. The paranoia regarding ego also is a named symptom, as is having what appears to be an ego the size of Mars, in truth he is quite terrified others will detect his deep anxieties, so he overcompensates by being arrogant and bullying others, and generally trying to sound like an authority by employing informal fallacies. His dishonesty, fwiw, is intentional, is known as gaslighting, and it is another common symptom of his illness.
1) Once again I will state that, and it should be obvious, we are not talking self discharge, we are talking permanent capacity loss. Those permanent capacity loss experiments are not tightly controlled for humidity, they are to represent real world storage conditions.
Once again, I will say permanent capacity loss only occurs to damaged cells. I was hoping you would explain exactly what damage caused the permanent capacity loss, but moisture such as found in a freezer condensing on the cell can do that. But i'll ask again, just what the hell are you talking about? Why would freezing prevent permanent capacity loss, and what causes the permanent capacity loss in the first place?
2) Even though we are talking permanent capacity loss, lets poke yet another hole in Chillin. The heat capacity of lithium ion cells is about 1034 joules/kg-1/c-1. I.e. 1034 joules for every degree C drop per kg of weight. It is an approximation. Lithium cells don't "freeze" at -18C as there is nothing to go through a phase change at those temps.
Straw man fallacy. I was speaking colloquially, not suggesting that the cell freezes the way water freezes solid, and I specifically addressed this before you showed up, that I only meant cold at a low enough temperature like 0°F such that if rapidly heated up could succumb to thermal shock.
From 22C to -18C, a 21700 cell, 0.07kg, requires 2900 joules to be removed. I could point out that a freezer is a form of heat pump, and does not actually require 2900 joules of energy to accomplish this but what would be the point?
I'd really like to know what you're getting at here, other than just to make pointless irrelevant babble. But think you've forgotten the part about refrigerating it for years.
A 21700 cell, nominal 3.6V, 5AH, has 65,000 joules of stored energy. Chalk up another wrong for Chillin.
Now what are you saying, falsely claiming I made some claim that I never made? Nice gaslight, as though no one could figure that out. Your calculations are incomplete, thus your conclusion is incorrect. First of all, you can't just pull calories out of something, In the real world, you're going to need a compressor and coolant and electricity to drive it, maybe something called a freezer, to actually cool a cell below freezing. You can't just wave your hand and show precisely what that energy is, and voila! Cold beer! You actually need the refrigerator, which will use a shitton more than 65K J to freeze it and keep it frozen for, say, a decade or so like your Trustfires.
3) If I was this grossly wrong, I would probably try to have less attitude while doing it.
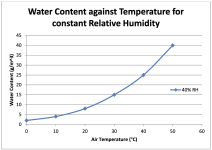
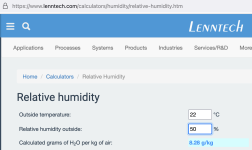
- (22C)/50% relative humidity air has 8.2g/kg of moisture. I.e. 8.2g or moisture (water) per kg or air. That is a lot of air by the way.
- (-13C)/100% relative humidity air has 1.8g/kg of moisture.
This is neither here nor there though. We already placed the batteries in the bag.
Yes, the magical bag that is a perfect insulator and doesn't obey the laws of thermodynamics. I love that bag.
As the temperature drops when the bag is placed in the freezer, the moisture will start to precipitate out. It will both precipitate out and form ice crystals that simply fall, and also freeze onto the surface of the first thing cold, which will be the plastic bag as it has less thermal mass than the battery and will cool faster. This will continue till you hit some equilibrium < 100% RH, probably closer to 50% RH, so you will get down to about 0.9g/kg of moisture in the air worst case when the freezer gets warmer at least inside the bag and realistically less as the air inside the bag will experience a smaller swing due to the thermal mass of the battery which acts like an ice cube in the sealed air, with Brownian motion enough to achieve some homogeneity of the air.
So ...... what do you think is worse, 0.9g/kg of moisture at -18 to -13C or 8.2g or far worse at 22C?
You are asserting an awfully specific amount of moisture there, and an amount that seems low, almost as though your cognitive dissonance invented it in order to self fulfill your own prophecy. He's actually pulling fictional numbers out of his giant *** now and passing it off as technical knowledge. Impressive! You must be really smart to be able to make things up like that.
Normally we are not concerned with air moisture because it does not do much till very high.
Except when things are very sensitive to moisture, it doesn't take a lot. But there in fact is a lot of moisture, up to 80%-100% for several hours at a time until the temperature drops again, at which point a lot of that vapor condenses on everything, including inside magical Ziploc bags that were not evacuated of air.
We are concerned with it condensing as liquid water dissolves impurities and can create conductive paths.
And you know what else? the water makes the air more conductive.
However, we are at -13 to -18C, and for the cell, typically much closer to -18C. There is no liquid water to dissolve impurities in solution. There is only ice. The very small amount of moisture in the bag will go from ice to vapour without a liquid phase as the temperatures are too low once it all freezes.
Nice fairy tail. I love how you assert that there will be perfect and predictable order in a chaotic system of oscillating tempertures and unknown amounts of water being added to the system every time the door is opened.
The ziploc or tupperware is essentially air tight, so adding external moisture has no appreciable effect on what is inside the bag.
But the moisture in the bag or Tupperware will condense and vaporize as the temperature changes because plastic is a poor thermal insulator and can not prevent it from occuring,
You may get ice forming on the outside of the bag since it is cold but most of that sublimates away as the RH drops once the freeze cycle restarts and the Rh drops.
What you've done is used false reasoning to sound scientific to try to persuade what I must now call victims of your narcissism to damage their cells by storing them in the freezer for no reason.
See, this is the difference between reading about stuff on the web, and say working in industry and having a relevant educational background so that you can tie real world experience to the underlying physics.
See, this is just another fallacy called appeal to authority, used in conjunction with ad hominem, so another fallacy. I think it is fitting Elvis ended his nutty argument on two fallacies. Buttons it up nicely.