There is some potentially confusing information being passed here that I'd like to try to clarify.
There are two kinds of battery capacity losses -- permanent and non-permanent (or recoverable). The latter is basically "self-discharge" (can also include parasitic discharge from something like a protection circuit, but that is typically a very small drain on the order of micro amps) and you reverse it by recharging the cell.
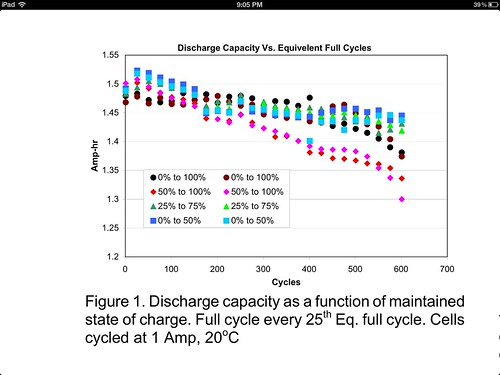
Permanent capacity loss is not recoverable by recharging or other means. It's a degradation arising from use and aging (the cells don't even have to be used). It is mainly due to the number of full charge/discharge cycles, battery voltage and temperature. All of these factors are more stressing on the cell and wear it out faster.
In your case, you are asking about proper cell storage in the flashlight. You want the flashlight to be immediately ready to use, yet you are also concerned about longevity of the cells, and presumably that the cells haven't self-discharged so much that you have no capacity remaining when you need to use the light. Ideally, you'd like to store the batteries at full charge for max readiness and run time, but the longer a Li-ion cell is exposed to high voltage (e.g., above 4.10V), the more permanent loss the cell suffers.
The generic Li-ion data says that if you store the cell at full charge (i.e., 4.20V resting voltage) at 25C (77F), you will permanently lose about 20% capacity per year. If you store the cell at an initial state of charge of 40% full at 25C, then you will permanently lose about 4% capacity per year. On top of that permanent capacity loss, there is the recoverable loss. Table 3 in the Battery University link provides recoverable (self-discharge) loss data. However, I think it is overly pessimistic. If you search CPF, IMO you'll find that the consensus is that *high-quality* Li-ions have very little self-discharge. So, IMO the only factor that you need to consider is what permanent capacity loss rate are you willing to tolerate.
The usual advice is to store Li-ions at 40% state of charge at 15C (60F). Should be no issues storing in the fridge (I'd probably not store them in the freezer because of condensation issues when re-exposed to ambient). Both types of capacity losses increase exponentially with temperature, so you definitely want to avoid exposing the cells to elevated temps.
The problem with 40% SoC is that it is only 40% SoC. Is that enough for your purposes? If the flashlight is an emergency light for which you might need days of run time, then maybe 40% is not enough.
Maybe go with full charge (4.20V) or 90% charge (4.10V) but store your loaded flashlight in the fridge, which is probably at about 4C (40F)?
Or look into a different battery chemistry like NiMH Eneloops or lead-acid.